Merck Zoztavax shingles vaccine could be a US$1 Bn. seller
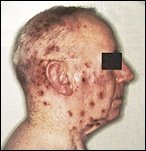 Had chicken pox ? 90% of people do, ususually in childhood, if so you could develop shingles later in life. Of the approximately one million people in the USA who get shingles each year, nearly half are over 60 that's 50 Mn Americans. More from VZW Foundation here
Had chicken pox ? 90% of people do, ususually in childhood, if so you could develop shingles later in life. Of the approximately one million people in the USA who get shingles each year, nearly half are over 60 that's 50 Mn Americans. More from VZW Foundation here
A market that can pay for a pharma product that has minimal impact on public health anywhere but will a shot in the arm for Merck sales and profits ... perhaps they will use the proceeds on a vaccine for malaria ?
The Shingles Prevention Study, was 5.5-year-trial randomized 19,270 US patients to and active herpes vaccine. The vaccine reduced the burden of illness due to herpes zoster by 61.1% (P<0.001), href="http://www.zostavax.com/clinical" data="">S Food and Drug Administration (FDA) review of the ZOSTAVAX concluded that the vaccine was effective at curbing shingles pain but did not significantly reduce either shingles-related hospitalizations or death.ZOSTAVAX is not a treatment for shingles or postherpetic neuralgia.
Today the FDA have approved a Merck shingles vaccine Zostavax (zoster vaccine live) for prevention of herpes zoster in patients 60 and older (although Merck applied for those aged 50 and over) - The vaccine will not be used for people who have had or now have shingles. Neither will people with weakened immune systems nor HIV patients (who are especially at risk) get the vaccines. Merck said it is ready to accept physician orders, with rapid shipment promised. (Merck Press release here)
Merck will perform a Phase 4 (postmarketing) study to provide additional safety information.Effectivley ZOSTAVAX is a 14 times strength child vaccine so should prove to be very safe in use.
In a statement issued by the FDA, Jesse L. Goodman, M.D., M.P.H., director of FDA's Center for Biologics Evaluation and Research, said the vaccine is "an important tool that can help prevent an illness that affects many older Americans and often results in significant chronic pain."
"It's a breakthrough in that it's the first vaccine that is actually
designed to keep an infection in check," said Dr. Walter Orenstein,
associate director of the Emory Vaccine Center at Emory University, who has
consulted for Merck.
Merck said the catalog price of the vaccine is US$145.35 for a 10-pack of single-dose vials of lyophilized vaccine with sterile diluent and $152.50 purchased as a single-dose vial of vaccine with sterile diluent. A singler dose should provide immunity for 4 years.
Merck is expected to gain approval next month for a cervical cancer vaccine (Gardasil) against the Human Papilloma Virus, that is likely to become popular among teenagers and young adults. Analaysts hope to see this as a US$2MN. seller and be mandated fro schoolchildren to prevent 1/2 Mn cervical cancer deaths annually.
Zostavax could be a US $1 billion's product for Merck per year by the end of this decade.
An der NYSE geben Merck&Co aktuell 0,5 Prozent auf 35,25 Dollar ab.
Merck needs some blockbusters. Vioxx in a global settlement could cost more than US$30 billion.
More bad news in the pipleine is the U.S. patent on Merck's popular cholesterol-lowering drug Zocor, 2md best seller in the US behind rival Lipitor, expires in 2006. The introduction of a generic copy of Zocor by mid-2006 could cut
in half Zocor's current annual U.S. sales of $4.5 billion.
Add this to the US $2.5 billion in annual sales sucked out of the company after Merck pulled Vioxx from the market.

3 comments:
I have worked for Merck in the past. They have massive research programs investigating diseases that primarily affect the aged and when I left in 2000 they had some interesting stuff on the way. I think they have a promising future.
They will pull through. the Gardasil product looks v. promising. However the UK NHS will still spend zillions on mass screening for cervical cancer with little effect.
Vioxx was a blunder - knowledge of problems was widespread and they hung in, hoping nothing would happen.
does anyone have information on zostavax?
Post a Comment